Immune cell implicated in development of lung disease following viral infection
Findings could help explain how asthma, COPD, severe COVID-19 are triggered
 Holtzman Lab
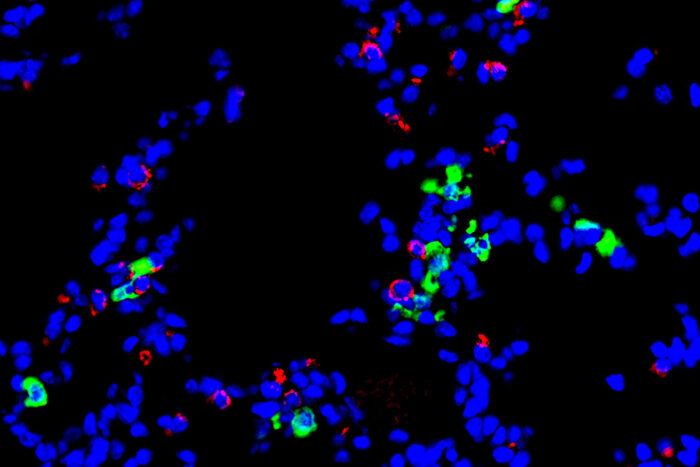
Holtzman LabA new study from Washington University School of Medicine in St. Louis implicates a type of immune cell — called a dendritic cell — in the development of chronic lung diseases that can follow after a respiratory viral infection. Shown is a stained section of mouse lung. Epithelial cells, which line the airway, are red. Dendritic cells are green. Any cell nuclei are blue.
Scientists at Washington University School of Medicine in St. Louis have implicated a type of immune cell in the development of chronic lung disease that sometimes is triggered following a respiratory viral infection. The evidence suggests that activation of this immune cell — a type of guardian cell called a dendritic cell — serves as an early switch that, when activated, sets in motion a chain of events that drives progressive lung diseases, including asthma and chronic obstructive pulmonary disease (COPD).
The new study, published in The Journal of Immunology, opens the door to potential preventive or therapeutic strategies for chronic lung disease. More immediately, measuring the levels of these dendritic cells in clinical samples from patients hospitalized with a viral infection, such as influenza or COVID-19, could help doctors identify which patients are at high risk of respiratory failure and death.
Studying mice with a respiratory viral infection that makes the animals prone to developing chronic lung disease, the researchers showed that these dendritic cells communicate with the lining of the airway in ways that cause the airway-lining cells to ramp up their growth and inflammatory signals. The inflammation causes airway-lining cells to grow beyond their normal boundaries and turn into cells that overproduce mucus and cause inflammation, which in turn causes cough and difficulty breathing.
“We’re trying to understand how a viral infection that seems to be cleared by the body can nevertheless trigger chronic, progressive lung disease,” said senior author Michael J. Holtzman, MD, the Selma and Herman Seldin Professor of Medicine. “Not everyone experiences this progression. We believe there’s some switch that gets flipped, triggering the bad response. We’re identifying that switch and ways to control it. This work tells us that this type of dendritic cell is sitting right at that switch point.”
Holtzman’s past work had implicated the lining of the airway — where the viral infection takes hold — as the likely trigger for this process.
“But this study suggests that the cascade starts even further upstream,” said Holtzman, also director of the Division of Pulmonary and Critical Care Medicine. “Dendritic cells are telling the cells lining the airway what to do. There’s more work to be done, but this data tells us that the dendritic cells play an important role in getting the airway-lining cells onto the wrong path.”
Holtzman calls this dendritic cell a type of sentinel because its job is to detect an invading virus and trigger the body’s initial immune response against the infection. The problem comes when the cell doesn’t shut down properly after the threat has passed.
“Many people never develop chronic lung disease after a viral infection,” Holtzman said. “But others have a genetic susceptibility to this type of disease. People who are susceptible to virus-triggered disease include patients with asthma, COPD, and viral infections such as COVID-19. It’s really critical to look for ways to fix this disease response and prevent the problems that might occur after the virus has gone.”
In the meantime, Holtzman said, high levels of these dendritic cells and their products in the lungs of hospitalized patients could serve as a warning to doctors that such patients are likely to develop severe disease and should be provided with respiratory interventions and other supportive therapies that are precisely tailored to their disease process.
“Similarly, if this process is not underway, the patient might be more likely to avoid these types of long-term problems,” Holtzman said. “We’re pursuing this line of research to help improve prediction of severe lung disease after infection and to provide companion therapies that could prevent this switch from being flipped or flip it back to reverse the disease.”